Regulated doesn't mean manual.
FDA portals, clinical trial systems, and pharmacovigilance platforms — automated with full audit-ready compliance at every step.
Your Operations

FDA portals, clinical trial systems, pharmacovigilance platforms.

Tightrope

Record the process. AI builds, runs, and maintains it.

The Result
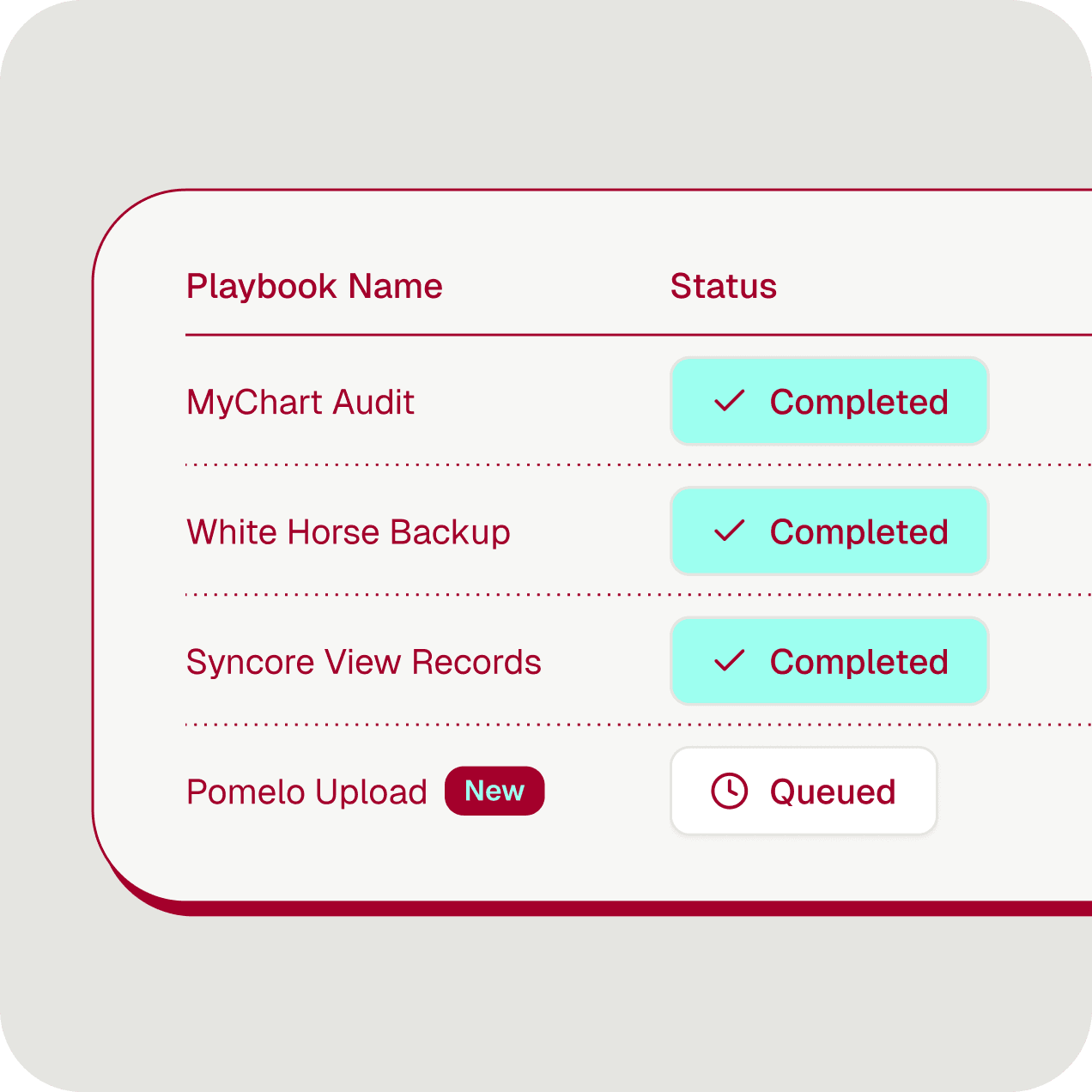
Faster submissions. Full audit trail. Days, not months.

What if your regulatory team could focus on decisions instead of data entry?
What if every step was logged, auditable, and your team could go from recording to production in days — not five months?
Automation discovery — as you work
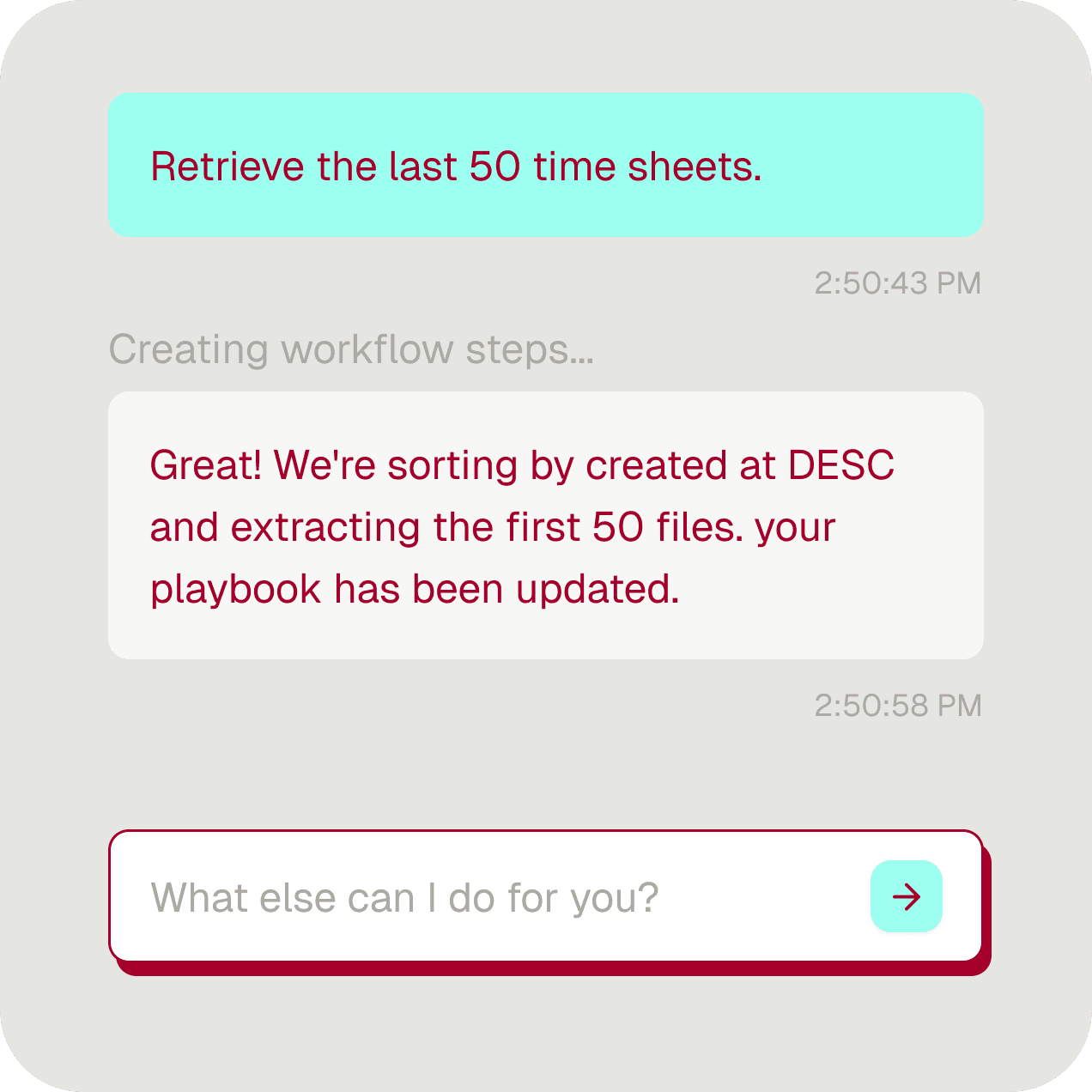
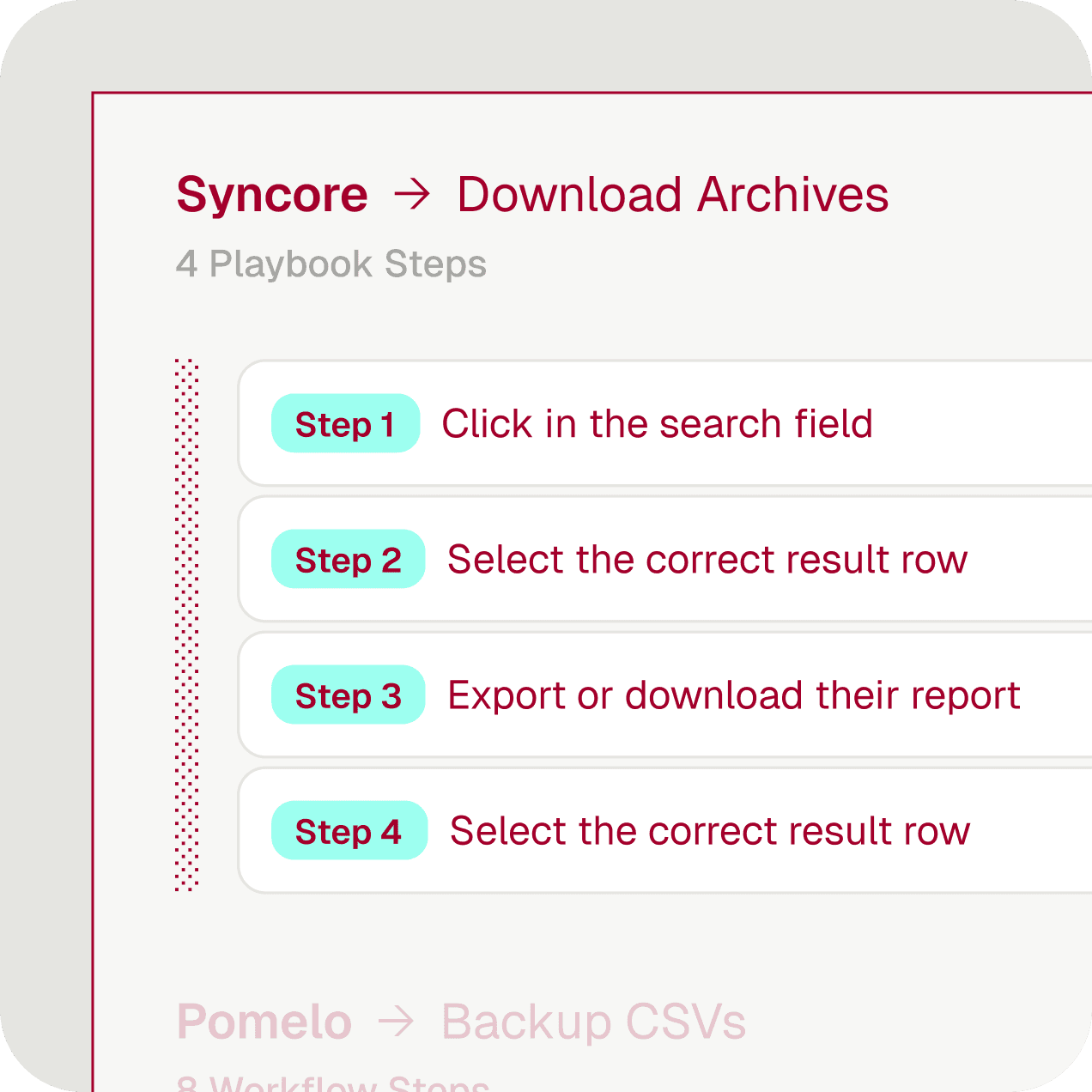
Record your process once. Tightrope identifies every step, every decision point, every exception.
No process mining tool required — discovery happens when you record
Your process expert drives — no consultants documenting workflows for months
Complex auth handled for you — 2FA, SSO, rotating credentials
Implementation at the speed of AI
AI turns the recording into a production automation. Deploy across customers, configure per account.
Zero to 100s of workflows a year — no technical team required
One playbook, many configurations — same process, different customers
Version control and deploy — treat automations like any other codebase
Self-healing automations built for the real world
No maintenance teams. No tickets. It stays running.
Self-healing — when portals change, the automation adapts instead of breaking
Full logs and visibility — every run logged, every action replayable, audit-ready
Scales without scaling the team — five automations or five thousand, same overhead